Technical studies of the performance of UTEM
Systematic studies were carried out to explore the potentials and limits of stroboscopic UTEM and single-shot DTEM
1. Stroboscopic UTEM
Image resolution with photoelectrons:

Lattice images with an information limit below 0.23 nm can be obtained with photoelectrons (repetition rate 2 MHz, total exposure time: 10 s). However, the actual image resolution is limited by the influence of the pump laser on the object.
EELS resolution with photoelectrons:
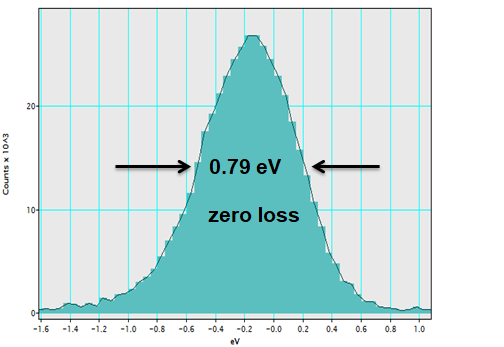
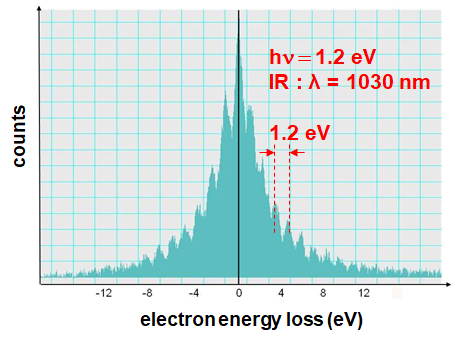
Synchronization of pump and probe pulses with photon-induced near field electron microscopy (PINEM):
Energy gain/loss of electrons in the photon field around the object:
Tradeoffs between temporal and energy resolution at different Wehnelt bias settings:
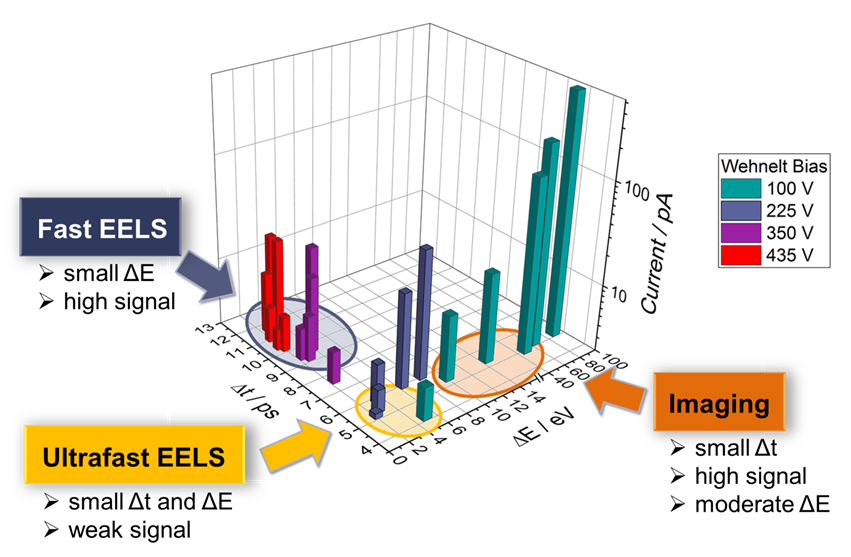
A temporal resolution of approximately 1 ps can be achieved with weak electron pulses.
Publication:
K. Bücker, M. Picher, O. Crégut, T. LaGrange, B. W. Reed, S. T. Park, D. J. Masiel, F. Banhart, The electron dynamics in an ultrafast transmission electron microscope with Wehnelt electrode, Ultramicroscopy, 171, 8-18 (2016)
2. Single-shot DTEM
Image resolution with single photoelectron pulses:

Quantitative core-loss EELS with single photoelectron pulses:
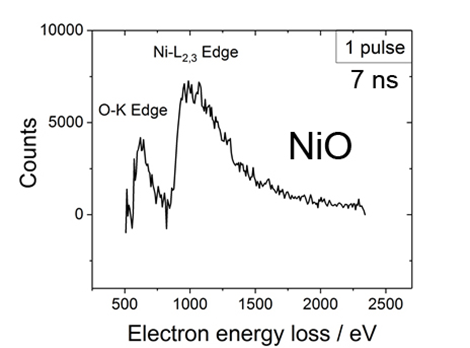
The pump laser can be focused onto a spot of 100 µm on the specimen. This allowed us to do up to 150 single-shot experiments on one specimen grid, although the transformation is irreversible.
Publications:
M. Picher, K. Bücker, T. LaGrange, F. Banhart, Imaging and electron energy-loss spectroscopy using single nanosecond electron pulses, Ultramicroscopy, 188, 41-47 (2018)
M. Picher, S. K. Sinha, T. LaGrange, F. Banhart, Analytics at the nanometer and nanosecond scales by short electron pulses in an electron microscope Chem Texts 8, 18 (2022)
Applications in materials science
1. Nanosecond quantitative analysis during chemical reactions in nanocrystals
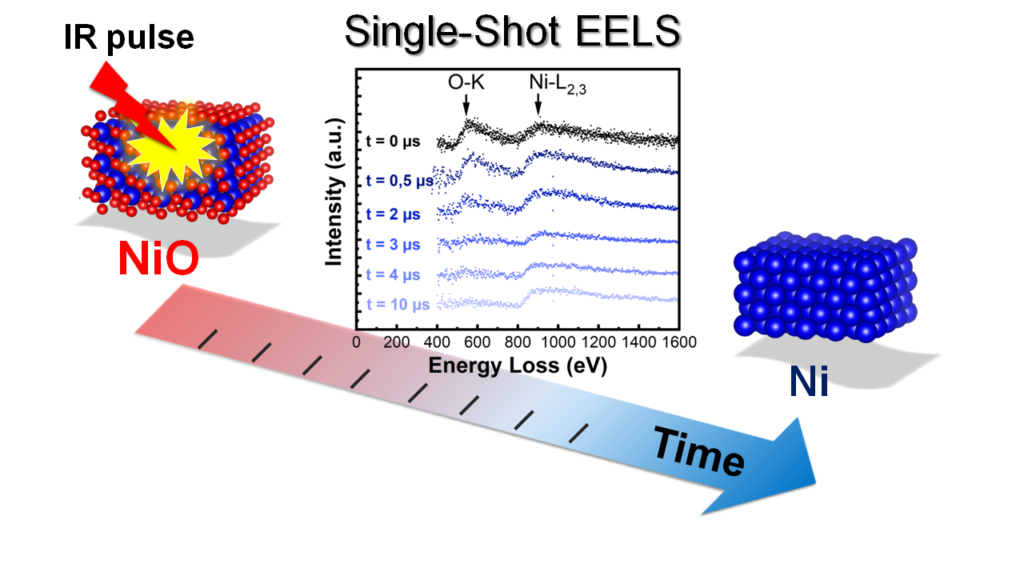
Chemical reactions in nanocrystals are difficult to study due to the small size of the reactants and the very short reaction times. At high temperatures, it might take only microseconds or even nanoseconds until a reaction front has traversed a nanometer-size crystal. Nevertheless, reaction kinetics in nanocrystals is highly important, e.g., in catalysis where the kinetics of oxidation or reduction of nanoparticles is essential for optimizing the catalytic action under extreme conditions. Reaction constants, activation energies or the presence of short-lived transition states are often unknown. Many solid-state reactions in nanomaterials are irreversible by nature, so the reaction cannot just be repeated to sum up signal emerging from the small objects.
DTEM (dynamic TEM) is the single-shot approach of (ultra-)fast TEM. Analytical DTEM, giving access to the elemental composition of nanomaterials with nanometer spatial and nanosecond time resolution, was developed at the IPCMS. The UTEM is used to study irreversible chemical reactions with nanometer and nanosecond precision. In a pump-probe approach, chemical reactions are induced thermally by an intense laser pulse and studied after an adjustable delay with a nanosecond electron pulse in the TEM.
Although the mutual repulsion of electrons within the pulses still limit spatial, temporal and energy resolution, combined imaging, diffraction and EELS studies turned out to be possible so that a comprehensive picture of the reaction kinetics in nanocrystals can be obtained. We have demonstrated this technique in a study of the fast reduction of nickel oxide nanocrystals at high temperature. The combination of imaging, electron diffraction and EELS gives unprecedented insight into the dynamics of the reaction. The kinetics of the reaction, in particular the reaction order and rate constant as well as the presence of liquid nickel as a transition state, allows the detailed understanding of the reaction mechanisms. The new approach has the potential to replace the complicated combination of different characterization techniques such as diffraction in a synchrotron, optical spectroscopy and microscopy.
Publication:
K. Sinha, A. Khammari, M. Picher, F. Roulland, N. Viart, T. LaGrange, F. Banhart, Nanosecond electron pulses in the analytical electron microscopy of a fast irreversible chemical reaction, Nature Communications 10, Article 3648 (2019), https://www.nature.com/articles/s41467-019-11669-w
——————————————————————————————————-
2. Time-resolved studies of photoswichable spin-crossover nanomaterials
See our recent publications:
Y. Hu, M. Picher, N. M. Tran, M. Palluel, L. Stoleriu, N. Daro, S. Mornet, C. Enachescu, E. Freysz, F. Banhart, G. Chastanet: Photo-Thermal Switching of Individual Plasmonically Activated Spin Crossover Nanoparticle Imaged by Ultrafast Transmission Electron Microscopy, Advanced Materials 33, 2105586 (2021) , https://doi.org/10.1002/adma.202105586
Y. Hu, M. Picher, M. Palluel, N. Daro, E. Fresz, L. Stoleriu, C. Enachescu, G. Chastanet, F. Banhart, Laser-driven transient phase oscillations in individual spin-crossover particles, Small 2303701 (2023) https://onlinelibrary.wiley.com/doi/full/10.1002/smll.202303701
H. Mba, M. Picher, N. Daro, M. Marchivie, P. Guionneau, G. Chastanet, F. Banhart, Lattice defects in sub-micrometer spin-crossover crystals studied by electron diffraction, J. Phys. Chem. Lett. 14, 8100 (2023)
